Preparation of Blood Components From Whole Blood for Transfusion
A single whole blood donation can be separated into different components to provide treatment to more than one patient. One unit of whole blood can be broken down into one unit of packed red cells, one unit of platelets, and one unit of fresh frozen plasma/cryoprecipitate. This practice avoids wastage of collected whole blood (each component is stored at a temperature that is optimal for that component), allows administration of specific replacement therapy, and also avoids transfusion of unnecessary blood elements that are not required by the patient.
There are two methods for collection of blood for preparation of blood components:
- Single whole blood donation: Preparation of blood components has been greatly facilitated by the introduction of double and triple bags having closed integral tubing. After collection of a unit of whole blood in the primary bag, blood components can be separated from one another by differential centrifugation due to differences in their specific gravities. After their separation, various components can be transferred from one bag to another in a closed circuit thus avoiding exposure to the external environment and maintaining the sterility. Blood should be processed for component separation within 6 hours of collection (Figure 1198.1 and 1198.2).
- Apheresis: This is a procedure in which a suitable donor is connected to an automated cell separator machine (that is essentially designed as a centrifuge) through which whole blood is withdrawn, the desired blood component is retained, and the remainder of the blood is returned back to the donor. Depending on the component that is separated and removed, the procedure is called plateletpheresis, leukapheresis, or plasmapheresis.
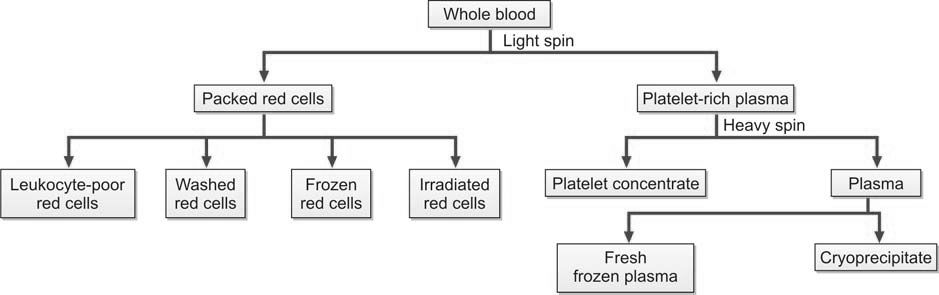
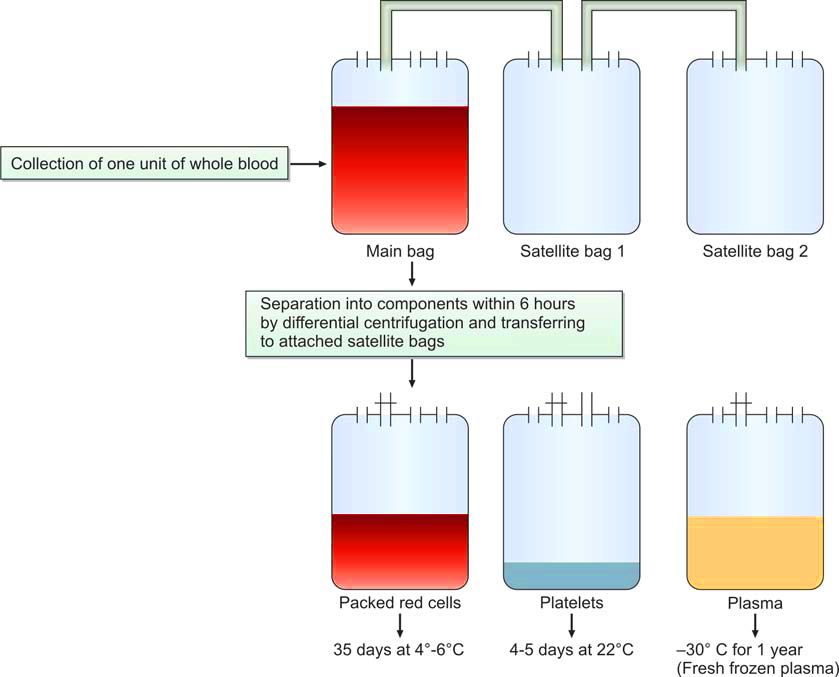
Terms used in transfusion therapy are shown in Table 1198.1.
| Blood product A therapeutic substance prepared from human blood |
| Whole blood One unit of non-separated donor blood collected in an appropriate container containing the anticoagulant-preservative solution |
| Blood component A constituent separated from whole blood by differential centrifugation or that is obtained directly from the donor by apheresis |
| Plasma derivative Human plasma proteins obtained from multiple donor units of plasma under pharmaceutical manufacturing conditions. These products are heat-treated or chemical-treated to inactivate lipid-enveloped viruses. |
Plasma derivatives like factor concentrates and immunoglobulins can also be prepared by recombinant DNA technology |
WHOLE BLOOD
Whole blood is one unit of donor blood collected in a suitable anticoagulant-preservative solution (citrate phosphate dextrose adenine or CPDA-1). Its total volume is about 400 ml (350 ml of blood + 49 ml of anticoagulant). It consists of cellular elements and plasma. Whole blood is stored in an approved blood bank refrigerator at 4°-6°C. The shelf life of such blood (collected in CPDA anticoagulant) is 35 days. It does not contain functionally effective platelets and labile coagulation factors (F V and F VIII). Transfusion of whole blood should commence within 30 minutes of removal from the refrigerator and should be complete within 4 hours of starting. Transfusion of one unit raises hemoglobin by 1 gm/dl or hematocrit by 3%.
Indications and contraindications for whole blood transfusion are given in Table 1198.2.
Indications
|
Contraindications
|
BLOOD COMPONENTS
Blood components are listed in Table 1198.3.
Cellular components
|
Plasma components
|
RED CELL COMPONENTS
1. Packed red cells:
Packed red cells are prepared by removing most of the plasma from one unit of whole blood (hematocrit 70-75%). Whole blood is either allowed to sediment overnight in a refrigerator at 2-6°C or is spun in a refrigerated centrifuge. Supernatant plasma is then separated from red cells in a closed system by transferring it to the attached empty satellite bag. Red cells and a small amount of plasma are left behind in the primary blood bag. Packed red cells have a high viscosity and therefore the rate of infusion is slow. Transfusion of one unit of red cells increases hemoglobin by 1 gm% (or increases hematocrit by 3%).
Indications for packed red cells are shown in Table 1198.4.
|
2. Red cells in additive solution (Red cell suspension):
These are red cells with minimal residual plasma and an additive solution (SAG-M which contains saline, adenine, glucose, and mannitol). This increases shelf life from 35 days to 42 days. After collection of whole blood in the primary collection bag (containing CPDA-1), maximum amount of plasma is removed (after centrifugation) and transferred to one satellite bag. The additive solution from the second satellite bag is transferred into the primary collection bag (containing packed red cells) in a closed system.
Indications for red cells in SAG-M are similar to those for packed red cells.
3. Leukocyte-poor red cells:
Leukocyte-poor red cells contain < 5 × 106 white cells per bag. Methods for leukocyte depletion are (i) leukocyte-reduction filters, and (ii) removal of buffy coat. Indications for leucocyte-poor red cells are: (i) prevention of HLA immunization in patients who are likely to receive allogeneic bone marrow transplantation, (ii) prevention of febrile nonhemolytic transfusion reactions in persons receiving multiple transfusions, and (iii) prevention of transmission of cytomegalovirus.
4. Washed red cells:
Red cells can be washed with normal saline to remove plasma proteins, white cells, and platelets. Such red cells are used for IgA-deficient individuals who have developed anti-IgA antibodies, as exposure will lead to anaphylaxis.
5. Frozen red cells:
If a cryoprotective agent such as glycerol is added, red cells can be stored frozen for upto 10 years. This method can be used for storage for donor red cells with rare blood groups, for future autologous transfusion, and for individuals who have repeated febrile nonhemolytic transfusion reactions.
6. Irradiated red cells:
Gamma-irradiation of red cells inactivates lymphocytes and prevents graft vs. host disease. Irradiated red cells are indicated for intrauterine or premature neonate transfusions, and in individuals with immunodeficiency, and in those receiving blood from first-degree relative donors.
PLATELETS
Platelet concentrates can be obtained from single donor units or by plateletpheresis.
1. Platelet concentrate (Random donor platelets prepared from whole blood unit):
One unit of whole blood is centrifuged (light spin) to obtain platelet-rich plasma (PRP). PRP is then transferred to the attached satellite bag and spun (high spin) to get platelets at the bottom and supernatant plasma. Most of the supernatant is returned back to the primary collection bag or to another satellite bag, leaving behind 50-60 ml of plasma with the platelets.
Platelets are stored at 20°-24°C with continuous agitation (in a storage device called platelet agitator). Maximum period of storage is 5 days.
One unit of platelet concentrate contains > 45 × 109 platelets. Transfusion of one unit will raise the platelet count in the recipient by about 5000/μl. The usual adult dose is 4-6 units of platelet concentrate (or 1 unit/10 kg of body weight). These units (which are from different donors) are pooled into one bag before transfusion. This dose will raise the platelet count by 20,000 to 40,000/μl.
2. Plateletpheresis (Single donor platelets):
In platelet pheresis, a donor is connected to a blood cell separator machine in which whole blood is collected in an anticoagulant solution, platelets are separated and retained, and remaining components are returned back to the donor. With this method, a large number of platelets can be obtained from a single donor (equivalent to 6 units of platelet concentrate). This method is especially suitable if HLA-matched platelets are required (i.e. if patient has developed refractoriness to platelet transfusion due to the formation of alloantibodies against HLA antigens). The usual indications and contraindications for administering platelets are shown in Table 1198.5.
Indications
|
Contraindications
|
PLASMA COMPONENTS
The main plasma components are fresh frozen plasma and cryoprecipitate.
1. Fresh frozen plasma (FFP):
FFP is prepared from whole blood within 6 hours of collection because after this time labile coagulation factors are lost. Plasma is separated from whole blood by centrifugation, expressed into the attached satellite bag, and rapidly frozen at –20°C or at lower temperature. FFP contains all the coagulation factors.
FFP can be stored for 1 year if temperature is maintained below –25°C. When required for transfusion, FFP is thawed between 30-37°C and then stored in the refrigerator at 2-6°C. Since labile coagulation factors rapidly deteriorate, FFP should be transfused within 2 hours of thawing.
Indications for FFP are shown in Table 1198.6.
|
2. Cryoprecipitate:
Cryoprecipitate is prepared from plasma that has been freshly separated (within 6 hours of collection) by rapidly freezing it at -20°C or lower and thawing it slowly at 4-6°C. A white flocculent precipitate and plasma are obtained. The mixture is centrifuged and supernatant plasma is removed leaving behind sediment of cryoprecipitate suspended in 10-20 ml of plasma. The unit is then refrozen (-20°C or colder) and can be stored at this temperature for 1 year. When needed, cryoprecipitate is thawed at 30-37°C, required donations are pooled and transfused to the patient. Cryoprecipitate contains F VIII, von Willebrand factor, fibrinogen, F XIII, and fibronectin. Indications for cryoprecipitate are F VIII deficiency (if F VIII concentrate is not available), von Willebrand disease, and deficiency of fibrinogen.
PLASMA DERIVATIVES
Plasma derivatives are manufactured by fractionation of large volumes of pooled human plasma. Important plasma derivatives are listed in Table 1198.7.
|
1. Human albumin solutions:
Albumin is prepared by cold ethanol fractionation of pooled plasma and is sterilized during manufacture to destroy viruses and bacteria. Albumin is used as a replacement fluid in therapeutic plasma exchange, and for treatment of diuretic-resistant edema of hypoproteinemia.
2. F VIII concentrate:
Freeze-dried F VIII concentrate is prepared by fractionation from large pools of fresh frozen plasma. To reduce the risk of transmission of viral infections, it is treated with heat or chemicals during manufacturing process. F VIII concentrate is the treatment of choice for treatment of hemophilia A and severe von Willebrand disease.
3. Prothrombin complex concentrate (PCC):
PCC contains factors II, VII, IX, and X, and also protein C and S. Main uses of PCC are (i) deficiency of F IX, (ii) deficiency of F VIII with development of inhibitors against F VIII, and (iii) inherited deficiency of factors II, VII, and X. A serious risk of PCC is thrombotic complications due to the presence of small amounts of activated coagulation factors.
4. Immunoglobulins:
Immunoglobulins are obtained by cold ethanol fractionation of large pools of human plasma. They are of two types: specific and nonspecific.
a. Non-specific immunoglobulins:
These are derived from the pooled plasma of non-selected donors. Some indications include (i) passive prophylaxis of viral infections like hepatitis, rubella, and measles, (ii) treatment of hypogammaglobulinaemia, (iii) autoimmune thrombocytopaenic purpura to induce a rise in platelet count, and (iv) neonatal sepsis.
b. Specific immunoglobulins:
They are obtained from donors who have selected high titer IgG antibodies. Anti-RhD immunoglobulin is prepared from plasma of Rh-negative donors who have produced anti-D following immunization; it is used for prevention of sensitization to RhD antigen in Rhnegative women giving birth to a Rh-positive baby. Other specific immunoglobulins include hepatitis B immune globulin, varicella-zoster immune globulin, and tetanus immune globulin that are used for passive prophylaxis of infections.
References
- World Health Organization. Blood Transfusion Safety: The Clinical Use of Blood. World Health Organization. Geneva, 2002.
- Comment
- Posted by Dayyal Dg.